
To harness the energy contained in Uranium, a nuclear reactor is used which, from a certain point of view, is designed according to the usual paradigm of energy production technologies: the heat produced by either uranium fission or coal combustion is used to heat water in a boiler and generate high pressure steam. In order to harness the energy contained in coal, it is burnt in thermal power plants, which also produces a lot of pollution in the extraction, transport, combustion and disposal of the smoke and ash. The precise value depends on many technical factors, but in essence, for the same weight, between three hundred thousand and three million times more nuclear energy is released from the fission of a given mass of uranium than is released - in the form of chemical bonding energy - by burning an equal mass of coal. One gram of uranium containing only 3% 235U can generate energy equivalent to that produced by burning 300 to 3,000 kg of coal. The fission of a single atom of 235U releases 202 million electronvolts (a unit used in electromagnetism and chemistry to measure the work done by an electron to cross a potential difference of 1 volt). It can only be stopped if we find a way to catch more stray neutrons than are formed and prevent them from hitting and breaking other 235U atoms. But that's not all: if there are many other 235U atoms surrounding the one that broke, the three neutrons produced by its fission can hit them and break them each of them will release three neutrons that in turn. The overcoming of the strong fundamental forces holding the two Barium and Krypton nuclei and the three neutrons together also releases a lot of energy in the form of kinetic energy. There are three neutrons missing, which have in fact broken away and shot off as three independent particles. But if we add up their total mass, we see that it comes to 141+92=233. 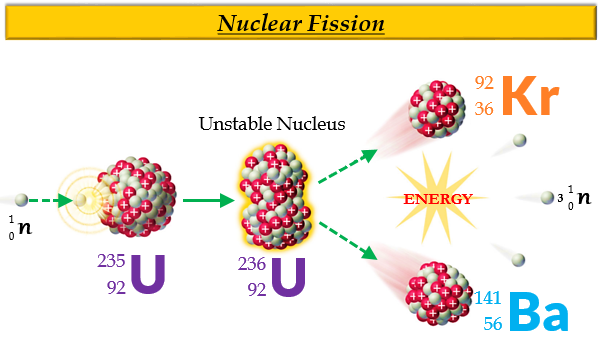
That makes a total of 92 and the proton count adds up. This process is called fission.īarium and Krypton have 36 and 56 protons respectively. It is still Uranium, because we already know that when you change the number of neutrons in an atom you do not change its nature, but the nucleus itself is no longer 235U: it becomes 236U, which reminds us that there is an extra neutron in it.Ģ36U, however, is terribly unstable: as soon as it forms, it falls apart, giving rise to a Barium 141Ba atom and a Krypton 92Kr atom. When the 235U atom is hit by a neutron with appropriate kinetic energy, a new isotope is formed. This isotope, remember, has its 92 protons, like all uranium atoms, but has "only" 235-92=143 neutrons. To achieve this, we must choose an isotope of an atom for example, the heaviest available on Earth, 235U. Nuclear fission occurs when a heavy nucleus breaks up and is split into two lighter ones, whose masses (when added together) are less than the original mass. Nuclear fission is a physical process in which the neutron is absorbed in the core of a heavy element and splits it into two lighter cores, converting part of the mass into energy.In terms of spelling there is only a different vowel and a consonant between them, but in reality these are two exactly opposite reactions in the atom. What is nuclear fission and how does it work? Thus, uranium 235 is the only naturally-occurring element fissile by thermal neutrons all other elements, such as the plutonium, are produced artificially. Before that, it was not known that a neutron could break a nucleus to two parts and release a very large amount of energy. Its main feature is that it is fissile with neutrons, which was accidentally discovered in 1938. It is considered one of the heaviest naturally-occurring elements as it is 18.7 times as dense as water. Uranium was discovered in 1789 by the German chemist Martin Klaproth and it was named after Uranus, the planet, which was discovered a few years earlier. What are the properties of the uranium atom? The most represented isotopes of uranium in nature are uranium 238 (99.29%) and uranium 235 (0.71%). The decay time of uranium is comparable to that of the earth, so it is still found in the Earth's crust. The released energy is stored in the Earth's crust and it contributes to the geothermal energy of our planet. :max_bytes(150000):strip_icc()/fission-of-a-uranium-nucleus-141483757-579266b03df78c173498d794.jpg)
Uranium is not a stable element and it gradually decays, which means that it emits energy. The most economic minerals are uraninite and carnotite and in low concentrations it can even be found in the sea. It can be found in an average concentration of 2 grams per tonne and in various mineral forms. Uranium is a chemical element to be found in the Earth's crust ever since the formation of our planet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
